The Surprising Truth About Cation: What It Is and Why It Matters More Than You Think 2026
Introduction
Have you ever wondered why your heart keeps beating rhythmically, why salt dissolves so easily in water, or how your muscles contract when you pick up a glass? The answer, surprisingly, comes down to something incredibly small — a cation.
A cation is a positively charged ion. It forms when an atom or molecule loses one or more electrons. That simple act of losing electrons changes everything — from how substances behave chemically to how your body functions biologically. Cations are literally everywhere: in your blood, in the soil, in the water you drink, and in the batteries powering your phone.
In this article, you’ll get a complete, easy-to-understand breakdown of what a cation is, how it forms, the different types, real-world examples, and why understanding cations can change the way you see the world around you. Whether you’re a student, a curious mind, or someone brushing up on chemistry, this one’s for you.
What Exactly Is a Cation?
Let’s start with the basics. A cation is an ion that carries a positive charge. The word itself comes from the Greek word kation, meaning “going down.” That name traces back to early electrochemistry experiments, where positively charged ions moved downward toward the negatively charged electrode called the cathode.
When an atom is neutral, it has an equal number of protons and electrons. But when it loses one or more electrons, the number of protons (positive charges) exceeds the number of electrons (negative charges). That imbalance creates a net positive charge — and that’s your cation.
Think of it like a tug-of-war. Electrons are the rope. When an atom lets go of that rope and loses electrons, the positive side wins. The result is a cation.
A Simple Way to Remember It
Here’s a trick I always find helpful: cation sounds like it starts with “cat” — and cats have claws that are positive (they scratch!). Meanwhile, anions are negative. It’s a silly memory trick, but it works surprisingly well.
How Does a Cation Form?
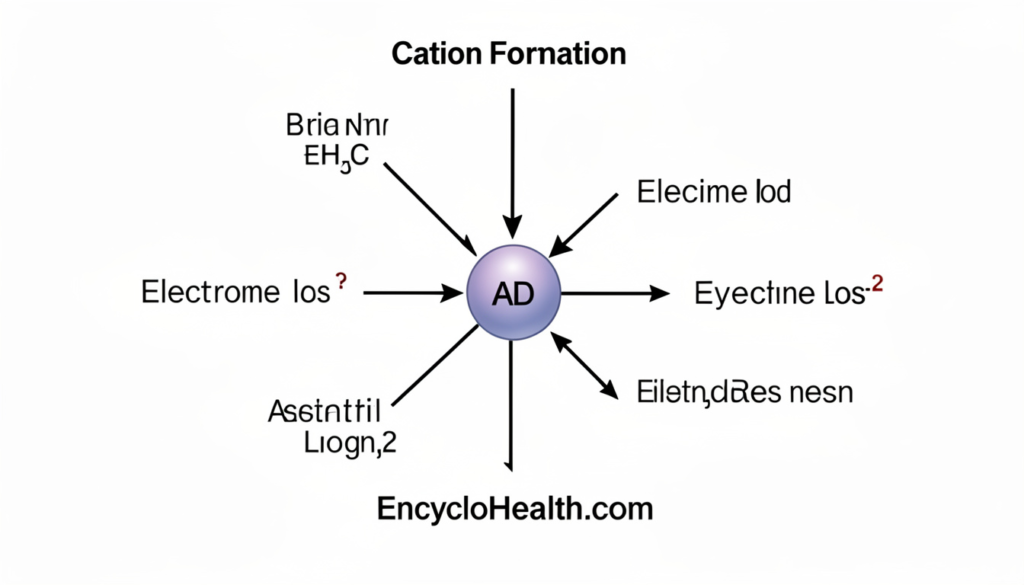
Cation formation is all about electron loss. This process is called ionization. Several factors drive an atom to lose electrons and become a cation.
Ionization Energy
Every atom holds onto its electrons with a certain grip — scientists call this ionization energy. Metals typically have low ionization energy. That means they give up electrons easily and almost always form cations. Sodium (Na), potassium (K), calcium (Ca), and magnesium (Mg) are perfect examples.
Non-metals, on the other hand, tend to have high ionization energy. They hold their electrons tightly and prefer to gain electrons instead, forming anions (negatively charged ions).
The Role of Chemical Reactions
Cations form during chemical reactions all the time. When sodium metal reacts with chlorine gas to form table salt (NaCl), sodium loses an electron and becomes Na⁺ — a cation. Chlorine gains that electron and becomes Cl⁻ — an anion. The two oppositely charged ions attract each other and form an ionic bond.
This is one of the most fundamental processes in all of chemistry.
Dissolution in Water
When ionic compounds dissolve in water, they split into their component ions. Table salt dissolving in water, for example, releases Na⁺ and Cl⁻ ions. The Na⁺ is your cation — floating freely in solution and ready to interact with other molecules.
Types of Cations You Should Know
Not all cations are created equal. They vary in charge, size, and behavior. Here are the main types.
Monovalent Cations (1+ Charge)
These cations carry a single positive charge. They’ve lost just one electron. Common examples include:
- Sodium (Na⁺) — critical for nerve function
- Potassium (K⁺) — essential for heart rhythm
- Hydrogen (H⁺) — defines acidity and pH
- Silver (Ag⁺) — used in photography and medicine
Divalent Cations (2+ Charge)
These have lost two electrons. They tend to be more reactive and play major biological roles.
- Calcium (Ca²⁺) — builds bones and triggers muscle contraction
- Magnesium (Mg²⁺) — activates hundreds of enzymes
- Iron (Fe²⁺) — carries oxygen in hemoglobin
- Zinc (Zn²⁺) — supports immune function and wound healing
Trivalent Cations (3+ Charge)
These cations have lost three electrons and tend to form strong ionic bonds.
- Aluminum (Al³⁺) — used in water purification
- Iron (Fe³⁺) — involved in electron transfer in biochemistry
- Chromium (Cr³⁺) — plays a role in glucose metabolism
Polyatomic Cations
Not all cations come from single atoms. Some are groups of atoms that collectively carry a positive charge.
- Ammonium (NH₄⁺) — common in fertilizers and biological systems
- Hydronium (H₃O⁺) — found in acidic water solutions
- Nitronium (NO₂⁺) — used in organic chemistry reactions
Cation vs. Anion: What’s the Real Difference?
People often mix these two up. The difference is actually very straightforward once you know what to look for.
A cation carries a positive charge — it has lost electrons. An anion carries a negative charge — it has gained electrons. In any ionic compound, you’ll always find both: a cation and an anion balancing each other out.
In table salt (NaCl), Na⁺ is the cation and Cl⁻ is the anion. In calcium chloride (CaCl₂), Ca²⁺ is the cation and Cl⁻ is again the anion. The cation is always the metal (or metal-like) part of the compound, while the anion is usually a non-metal.
One more key difference: in electrochemistry, cations migrate toward the cathode (negative electrode), while anions move toward the anode (positive electrode). That’s actually how the names came about historically.
Cation Examples in Everyday Life
Here’s where things get genuinely interesting. Cations aren’t just textbook concepts. They show up in your daily life constantly.
In Your Body
Your body is essentially an electrochemical machine, and cations run much of the operation.
- Na⁺ and K⁺ work together in the sodium-potassium pump, a process that keeps every cell in your body alive. Without this pump, your neurons couldn’t fire and your muscles couldn’t move.
- Ca²⁺ triggers muscle contractions — including your heartbeat. Every time your heart beats, calcium cations rush into muscle cells.
- Mg²⁺ activates over 300 enzyme reactions. Low magnesium is linked to fatigue, muscle cramps, and even anxiety.
- H⁺ (protons) determine whether your blood is too acidic or too alkaline. Even tiny shifts can be life-threatening.
In Drinking Water
Water quality is closely tied to cation content. Hard water, which is common in many regions, contains high levels of Ca²⁺ and Mg²⁺ cations. That’s why you get limescale on your kettle and why your soap doesn’t lather as well in hard water areas.
Water softeners work by swapping those calcium and magnesium cations for sodium cations through a process called ion exchange — a direct application of cation chemistry.
In Soil and Agriculture
Plants absorb nutrients from soil largely in the form of cations. Nutrients like K⁺, Ca²⁺, Mg²⁺, and NH₄⁺ are all cations that plants pull up through their roots. Soil scientists measure Cation Exchange Capacity (CEC) — a key indicator of how well soil can hold and supply these nutrients. Healthy, fertile soil has a high CEC. Poor soil has a low one.
Farmers and gardeners who understand cation exchange can dramatically improve crop yields just by managing their soil’s ionic balance. That’s pretty powerful knowledge for something that seems like a purely academic chemistry topic.
In Batteries and Electronics
Lithium-ion batteries — the kind in your phone, laptop, and electric car — work by moving Li⁺ cations back and forth between electrodes. When you charge your device, lithium cations migrate one way. When you use it, they migrate back. The movement of these cations is literally what powers your screen right now.
In Medicine
Cations are central to many medical treatments.
- Lithium cations (Li⁺) are used as a mood stabilizer for bipolar disorder.
- Platinum-based cations form the backbone of chemotherapy drugs like cisplatin.
- Iron cations are given as supplements to treat anemia.
- Calcium cations are administered intravenously in emergencies involving heart rhythm problems.
The Chemistry of Cations: Ionic Bonds and Solutions
Ionic Bonding
When a cation and an anion come together, they form an ionic bond — one of the strongest types of chemical bonds. The attraction between opposite charges holds these ions tightly together in a crystal lattice structure. Table salt’s rigid cubic crystal is a perfect example of this structure.
Ionic compounds formed this way have predictable properties: they tend to be solid at room temperature, have high melting points, and dissolve in water to conduct electricity.
Cations in Solution: Electrolytes
When cations dissolve in water, they become part of what’s called an electrolyte solution — a liquid that can conduct electricity. Sports drinks are marketed on this principle. They contain Na⁺, K⁺, Ca²⁺, and Mg²⁺ to replenish the electrolytes your body loses through sweat.
Electrolyte imbalances are genuinely dangerous. Hyponatremia (too little Na⁺) can cause brain swelling. Hypokalemia (too little K⁺) can trigger dangerous heart arrhythmias. These aren’t just chemistry facts — they’re life-and-death clinical realities.
Hydration of Cations
Something fascinating happens when a cation enters water. Water molecules, which are polar (partially charged), cluster around the cation with their negative oxygen ends pointing toward the positive ion. This is called the hydration shell. Smaller cations with higher charges tend to have tighter, more organized hydration shells — and this affects how they behave in biological systems.
Cation Exchange Capacity: Why It Matters in the Real World
If you’re into gardening, farming, or environmental science, you’ll encounter Cation Exchange Capacity (CEC) often.
CEC measures how many cations a soil can hold and exchange. Clay soils and soils rich in organic matter typically have high CEC values. Sandy soils have low CEC. A higher CEC means the soil can store more nutrients and release them steadily to plant roots.
Farmers use CEC data to make smarter decisions about fertilization. Environmental engineers use it to understand how contaminants (which often travel as cations) move through soil and groundwater. It’s one of the most practically useful applications of cation chemistry outside a lab.
Cation in Analytical Chemistry: How Scientists Detect Them
Identifying and quantifying cations is a major part of analytical chemistry. Several techniques are commonly used:
Flame tests are one of the oldest and most visual methods. Different cations produce distinct colors when burned — Na⁺ gives a bright yellow flame, K⁺ gives violet, Ca²⁺ gives brick red, and Cu²⁺ gives green. It’s one of the most visually satisfying experiments in all of chemistry.
Ion chromatography separates cations in a solution based on how strongly they interact with a charged resin. It’s used in water quality testing, food safety analysis, and pharmaceutical quality control.
Atomic absorption spectroscopy (AAS) measures how much light specific cations absorb. It’s incredibly precise and widely used in clinical labs to measure trace metals in blood and urine.
Common Misconceptions About Cations
A few things trip people up when learning about cations. Let me clear them up.
Misconception 1: Cations are always metals. While metals most commonly form cations, hydrogen (H⁺) is a non-metal cation found in virtually every acid. Ammonium (NH₄⁺) is a polyatomic cation with no metals involved at all.
Misconception 2: Cations are harmful. Many essential nutrients are cations. Your body couldn’t survive without them. The dose and context matter — not the charge.
Misconception 3: More charge always means more reactive. Higher-charged cations do tend to form stronger ionic bonds, but reactivity depends on many factors including electron configuration, atomic radius, and the surrounding chemical environment.
Conclusion
Cations are one of those foundational concepts that, once you truly understand them, seem to pop up absolutely everywhere. From the rhythm of your heartbeat to the battery in your phone, from the fertility of farmland to the clarity of your drinking water — cations are quietly running the show behind the scenes.
The key takeaways are simple: a cation is a positively charged ion formed when an atom loses electrons. They come in different types — monovalent, divalent, trivalent, and polyatomic. They’re essential in biology, agriculture, environmental science, medicine, and technology. And understanding them isn’t just for chemists — it’s genuinely useful knowledge for anyone curious about how the world works.
So next time you reach for a sports drink after a workout, take a calcium supplement, or charge your phone, take a second to appreciate the tiny, positively charged particles making it all possible.
What surprised you most about cations? Drop your thoughts, share this with a science-curious friend, or revisit your high school chemistry notes with fresh eyes — you might be amazed at how much more sense it all makes now.
Frequently Asked Questions (FAQs)
1. What is a cation in simple terms? A cation is a positively charged ion. It forms when an atom loses one or more electrons, giving it more protons than electrons.
2. What is the difference between a cation and an anion? A cation has a positive charge (loses electrons), while an anion has a negative charge (gains electrons). In salt (NaCl), Na⁺ is the cation and Cl⁻ is the anion.
3. What are common examples of cations? Common cations include Na⁺ (sodium), K⁺ (potassium), Ca²⁺ (calcium), Mg²⁺ (magnesium), H⁺ (hydrogen), and Fe²⁺ (iron).
4. Are cations good or bad for your health? Many cations are absolutely essential for health — calcium for bones, potassium for heart rhythm, sodium for nerve function. Problems arise only when they’re too high, too low, or in the wrong place.
5. Why do cations move toward the cathode? Cations carry positive charges and are attracted to the negatively charged cathode in an electrochemical cell. That’s actually where the name “cation” originally came from.
6. What is cation exchange capacity (CEC)? CEC measures how many positively charged ions (cations) a soil can hold and exchange. Higher CEC generally means more fertile, nutrient-retaining soil.
7. How are cations formed? Cations are formed through a process called ionization, where an atom or molecule loses one or more electrons — typically during chemical reactions, dissolution in water, or exposure to heat or electricity.
8. Is hydrogen a cation? Yes. H⁺ (a hydrogen atom that has lost its single electron, leaving just a proton) is a cation. It’s the cation responsible for acidity in chemical solutions.
9. What is a polyatomic cation? A polyatomic cation is a group of atoms bonded together that collectively carry a positive charge. Ammonium (NH₄⁺) is the most common example.
10. How are cations used in medicine? Cations are used in many medical contexts — lithium cations treat bipolar disorder, platinum cations are used in chemotherapy, iron cations treat anemia, and calcium cations are used in cardiac emergencies.
Also Read : Risk of Rain 2 Lunar Items

